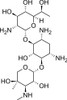
G418 Disulfate, EvoPure® is a highly pure (≥99.0%) version of G418 Disulfate. It has been purified to remove the impurities commonly present in G418 Disulfate. This aminoglycoside antibiotic, originally isolated from Micromonospora rhodorangea, is routinely used for gene selection in cell culture.
We also offer: