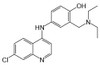
Amodiaquine Hydrochloride is a synthetic,4-aminoquinoline compound with anti-malarial and anti-inflammatory properties. It is a histamine-N-methyltransferase inhibitor that was developed in 1948. Amodiaquine Hydrochloride is a congener of chloroquine and its mechanism of action is similar. Its active metabolite is N-desethylamodiaquine.
Amodiaquine Hydrochloride is soluble in DMSO.
| Mechanism of Action | The mechanism of action for Amodiaquine is not entirely clear; however, it is thought to inhibit digestion of hemoglobin in malarial parasites, inhibiting heme polymerase activity. The result is an accumulation of free heme, which is toxic to the parasite. |
| Spectrum | Amodiaquine has a similar spectrum of activity as chloroquine (Plasmodium vivax, Plasmodium malariae) however; it has shown to be more effective against chloroquine-resistant Plasmodium falciparum. |
| Molecular Formula | C20H22ClN3O •2HCl •2H2O |
| Impurities | Individual Impurity: ≤0.5% Total Impurities: ≤1.0% |
| References |
Ahmad I, Ahmad T and Usmanghani K (1992) Amodiaquine Hydrochloride. In: Analytical profiles of drug substances and excipients. 21:43-73 Wennerholm et al (2006) Amodiaquine, its desethylated metabolite, or both, inhibit the metabolism of debrisoquine (CYP2D6) and losartan (CYP2C9) in vivo. Eur. J. Clin. Pharmacol. 62:539–546 |