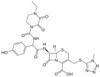
Cefoperazone Sodium is a third-generation, semi-synthetic, broad-spectrum cephalosporin antibiotic. It is resistant to β-lactamase and is often used to study compound-protein binding and expression and inhibition of penicillin binding proteins (PBPs).
| Mechanism of Action | Like β-lactams, cephalosporins interfere with PBP (penicillin binding protein) activity involved in the final phase of peptidoglycan synthesis. PBP’s are enzymes which catalyze a pentaglycine crosslink between alanine and lysine residues providing additional strength to the cell wall. Without a pentaglycine crosslink, the integrity of the cell wall is severely compromised and ultimately leads to cell lysis and death. Resistance to cephalosporins is commonly due to cells containing plasmid encoded β-lactamases. Cefoperazone however, is largely resistant to β-lactamases produced by resistant cells. |
| Spectrum | Cefoperazone is active against Gram-positive and Gram-negative bacteria (ie Pseudomonas aeruginosa). |
| Microbiology Applications | Cefoperazone is commonly used in clinical in vitro microbiological antimicrobial susceptibility tests (panels, discs, and MIC strips) against Gram-negative microbial isolates. Medical microbiologists use AST results to recommend antibiotic treatment options. Representative MIC values include:
For a representative list of Cefoperazone MIC values, click here. Media SupplementsCefoperazone can be used as a selective agent in several types of isolation media: Campylobacter Blood-Free Selective Agar - CCDA Selective Supplement Campylobacter Agar - Campylobacter Selective Supplement (Karmali) Blood Free Campylobacter Agar - CDMN Selective Supplement Bolton Broth - Bolton Broth Selective Supplement Campylobacter Agar Base - Modified Karmali Selective Supplement Bolton Broth - Modified Bolton Broth Selective Supplement |
| Molecular Formula | C25H26N9NaO8S2 |
| References | Brogden RN et al (1981) Cefoperazone: A review of its in vitro antimicrobial activity, pharmacological properties and therapeutic efficacy. Drugs 22:423-460 PMD 6459224 Hinkle AM, LeBlanc BM, Bodey GP (1980) In vitro evaluation of Cefoperazone. Antimicrobial Agents Chemother. 17(3):423-427 PMID 6448578 Matsubara N, Minami S, Muraoka T, Saikawa I, Mitsuhashi S (1979) In vitro antibacterial activity of Cefoperazone (T-1551), a new semisynthetic cephalosporin. Antimicrob Agents Chemother. 16(6):731-735 PMID 316988 |