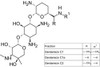
Gentamicin Sulfate, EP is an aminoglycoside antibiotic complex discovered in 1963 derived from fermentation of Micromonospora purpurea or Micromonospora echinospora. Gentamicin is composed of different components including Gentamicin C complex (Gentamicin C1, Gentamicin C1a, and Gentamicin C2) which makes up 80% of the compound and has the highest antibacterial activity, along with Gentamicin A, B, X, and a few others which make up the remaining 20% of Gentamicin and have lower antibiotic activity. Gentamicin Sulfate is suitable for use in cell culture to prevent and control bacterial contamination. The Centers for Disease Control (CDC) Standard Operating Procedure (SOP) DSR-052-04 includes the addition of Gentamicin Sulfate to viral transport media for SARS-CoV2 samples.
Gentamicin Sulfate, EP is soluble in water.
Gentamicin Sulfate, EP conforms to European Pharmacopoeia specifications.
We also offer:
- Gentamicin Sulfate, USP (G006)
- Gentamicin ReadyMadeTM Solution (G046)
- Gentamicin A Sulfate, EvoPure (G035)
- Gentamicin C1 Sulfate, EvoPure (G031)
- Gentamicin C1a Sulfate, EvoPure (G032)
- Gentamicin C2 Sulfate, EvoPure (G033)
- Gentamicin C2a Sulfate, EvoPure (G034)
- Gentamicin C2b Sulfate, EvoPure (G062)
- Gentamicin X2 Sulfate, EvoPure (G036)
| Mechanism of Action | Aminoglycosides are a widespread and versatile group of bioactive natural products. They target the 30S ribosomal subunit, blocking the translocation of peptidyl-tRNA from acceptor to donor. This results in an inability to read mRNA ultimately producing a faulty or nonexistent protein. |
| Spectrum | Gentamicin Sulfate, EP is a broad-spectrum antibiotic targeting Gram-positive and Gram-negative bacteria. It is effective against several strains of Mycoplasma. It also combats certain β-lactam sensitive VRE or vancomycin resistant Enterococcus; a "superbug." |
| Microbiology Applications | Gentamicin Sulfate is commonly used as a selective agent to select for cells containing the Gentamicin resistance gene, aacj-AaphD or aacC1. Gentamicin Sulfate is generally used at a concentration of 10 - 50 µg/mL for eukaryotic cells and 15 µg/ml for prokaryotic cells.
According to the CDC SOP (DSR-052-04) for Viral Transport Medium (VTM), Gentamicin Sulfate is used at a final concentration of 100 µg/ml. Media SupplementsGentamicin can be used as a selective agent in several types of isolation media: Columbia Blood Agar - Gardnerella vaginalis Selective Supplement VRE Medium - VRE Selective Supplement Burkholderia cepacia Agar Base - Burkholderia cepacia Selective Supplement Microfluidic droplet systems are popular analytical methods used in antimicrobial susceptibility testing (AST) to help study antimicrobial resistance (AMR). Droplet methods have many advantages over conventional AST methods. Authors used several antibiotics from TOKU-E including X to review the physicochemical properties to predict retention of antibiotics in droplets (Ruszczak et al, 2023). |
| Plant Biology Applications | Gentamicin Sulfate inhibited tracheary element differentiation in cultured explants of pith parenchyma from romaine lettuce (Lactuca sativa L. var. Romana) at 50 - 100 μg/ml. Similar results were obtained with cultured explants of Jerusalem artichoke tuber (Helianthus tuberosus L.). Callus formation was suppressed by Gentamicin Sulfate in both tissue systems. When studying cell division and xylem differentiation in plant tissue culture, concentrations up to 10 μg/ml is suggested. |
| Eukaryotic Cell Culture Applications | Gentamicin was found to inhibit Glucose-6-phosphate dehydrogenase (G6PD) enzyme activity in rat erythrocytes (Temel et al, 2018). A bovine macrophage (Bomac) cell line was used but proved to be contaminated with BVDV (bovine viral diarrhea virus). Both infected and cured cells were tested for uptake of Mycoplasma bovis in an in vitro model to dissect the molecular and cellular details of bovine mycoplasmosis using Gentamicin (TOKU-E) in a Gentamicin protection assay (Burgi et al, 2018). Gentamicin is an effective in vitro bacterial inhibitor that is nontoxic in tissue culture. It was non-toxic to RMK (Rhesus monkey kidney), HeLa, amnion, GMK, and WI-38 cell lines. It can also be used in virus tissue culture as it does not inhibit virus replication. (Rudin et al, 1970). When primary cultures of embryonic rat fibroblasts are exposed to Gentamicin, they develop typical lysosomal phospholipidosis characterized by decreased sphingomyelinase activity, increase in lipid phosphorus, and appearance of 'myeloid bodies' in lysosomes. However, when inhibitors of cysteine proteinases were used (leupeptin, E-64), authors observed a protective effect which could be due to increased sphingomyelinase activity (Montenez et al, 1984). |
| Cancer Applications | Ovarian melanoma tumor cells was studied in 3D culture and Gentamicin Sulfate was used to prevent contamination when studying ovarian cell lines (OVCAR3, SKOV3, 222, EG, and A2780-PAR ) and normal ovarian surface epithelial cell lines (HIO 1120 and HIO 180). Tumor cells formed matrix-rich tubular networks containing channels surrounding spheroids of tumor cells, and this network may represent either a primitive microcirculatory-like network, or a remodeled vascularized portion of a tumor (Sood et al, 2001). |
| Molecular Formula |
Gentamicin C Complex: C21H43N5O7 (C1); C19H39N5O7 (c1a); C20H41N5O7) (C2); C20H41N5O7 (c2a); C20H41N5O7 (c2b) |
| Molecular Weight | base: 477.59 (C1); 449.54 (C1a); 463.57 (C2); (463.57 (c2a); 463.57 (c2b) |
| Impurities |
Impurity A: Not more than 3.0% |
| Residual Solvents |
Methanol: Not more than 1.0%; |
| References |
Deng et al used Gentamicin Sulfate (TOKU-E) in Gentamicin protection assays to evaluate effect of miR-30c-1-3p and miR-30c-5p on intracellular Helicobacter pylori. Link to article. Ruszczak et al used Gentamicin Sulfate (TOKU-E) to study the physicochemical properties and predict retention of antibiotics in water-in-oil droplets for microfluidics. Link to article. References Bürgi N, Josi C, Bürki S and Schweizer, Pilo P (2018) Mycoplasma bovis co-infection with bovine viral diarrhea virus in bovine macrophages. Vet. Res. 49(1):2. PMID 29316971 Centers for Disease Control and Prevention. 2020. DSR-052-02: Preparation of viral transport medium. Link to SOP. Deng et al (2022) miR-30c increases the intracellular survival of Helicobacter pylori by inhibiting autophagy. Cellular Microbiol. 4536450 Gyetvai B et al (2015) Gentamicin Sulphate permeation through porcine intestinal epithelial cell monolayer. Act. Vet. Hungarica 63(1): 60-68 PMID 25655415 Kadurugamuwa JL, Clarke AJ and Beveridge TJ (1993) Surface action of Gentamicin on Pseudomonas aeruginosa. J. Bacteriol 175(18):5798-5805 PMID 8376327Martin NL and Bevridge TJ (1986) Gentamicin interaction with Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 29(6):1079-1087 PMID 2425732 Montenez JP, Kishore BK, Maldaque P and Tulkens PM (1984) Leupeptin and E-64, inhibitors of cysteine proteinases, prevent Gentamicin-induced lysosomal phospholipidosis in cultured rat fibroblasts. Toxicol. Lett. 73(3):201-208 PMID 8091428 Rudin A, Healey A, Phillips CA, Gump DW and Forsyth BR (1970) Antibacterial activity of Gentamicin Sulfate in tissue culture. Appl. Microbiol. 20(6):989-990 PMID 4992660 Sood AK (2001) Molecular determinants of ovarian cancer plasticity. Am. J. Pathol. 158(4):1279-1288. PMID 11290546 Temel Y, Ayna A, Shafeeq IH and Ciftci M (2018) In vitro effects of some antibiotics on glucose-6-phosphate dehydrogenase from rat (Rattus norvegicus) erythrocyte. Drug and Chemical Toxicol. DOI: 10.1080/01480545.2018.1481083 Wan J et al (1994) Intravesical instillation of Gentamicin Sulfate: In vitro, rat, canine, and human studies. Urology 43(4):531-536. PMID 8154077 |