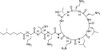
Polymyxin E1 Sulfate (syn: colistin A) is one of the two major components of Polymyxin E (colistin). Polymyxin E1 and E2 are structurally similar and differ only by a fatty acid group at the N-terminus. Polymyxin E1 contains 6-methyloctanoic acid and Polymyxin E2 (colistin B) contains 6-methylheptanoic acid. Together, Polymyxin E1 and E2 comprise approximately 85% of Polymyxin E; however, 13 different Polymyxin E components have been indentified.
We also offer:
- Polymyxin B Sulfate, USP (P007)
- Polymyxin B1 Sulfate, EvoPure® (P037)
- Polymyxin B1-I Sulfate, EvoPure® (P038)
- Polymyxin B2 Sulfate, EvoPure® (P039)
- Polymyxin B3 Sulfate, EvoPure® (P040)
- Polymyxin B6 Sulfate, EvoPure® (P054)
- Polymyxin E1 Sulfate, EvoPure® (P055)
- Polymyxin E2 Sulfate, EvoPure® (P056)
EvoPure® products are purified single antibiotic fractions, most compounds are >99% pure. Highly pure EvoPure® Polymyxin products can be used to analyze the specifc effects of individual Polymyxin B fractions.
| Mechanism of Action | Polymyxin E has a bactericidal effect on Gram-negative bacteria by interacting with and displacing essential ions in the lipopolysaccharide (LPS) outer cell wall leading to increased permeability and eventually lysis and death of the cell. |
| Spectrum | Polymyxin E is used primarily against Gram-negative bacteria including Pseudomonas aeruginosa, Klebsiella pneumoniae, and multi-drug resistant Enterobacteriaceae. |
| Microbiology Applications | Polymyxin E1 and E2 (colistin A and B, respectively) can be used individually to study and compare in vitro antimicrobial activity with Colistin (Polymyxin E complex) or other polymyxins. |
| Molecular Formula | C53H100N16O13 · xH2SO4 (lot specific) |
| References |
Falagas ME (2005) Colistin: The revival of polymyxins for the management of multidrug-resistant Gram-negative bacterial infections. Clinical Infect. Dis. 40(90):1333-1341 PMID 15825037 Bergen PJ et al (2006) Colistin Methanesulfonate is an inactive prodrug of Colistin against Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 50(6):1953-1958 PMID 16723551 Decolin D, Leroy P, Nicolas A, Archimbault P (1997) Hyphenated liquid chromatographic method for the determination of colistin residues in bovine tissues. J Chromatogr. Sci. 135(12):557-564 PMID 9397540 Leifert C, Ritchie JY and Waites WM (1991) Contaminants of plant-tissue and cell cultures. World J. Microbiol. Biotechnol. 7:452-469 PMID 24425131 Mueller MJ and Brodschelm W (1993) Signaling in the elicitation process is mediated through the octadecanoid pathway leading to jasmonic acid. Proc. Natl. Acad. Sci. USA 90:7490-7494
|